WESAIL new crown antigen saliva test kit into the Ministry of Commerce export white list
Release time:
2023-04-28 14:07
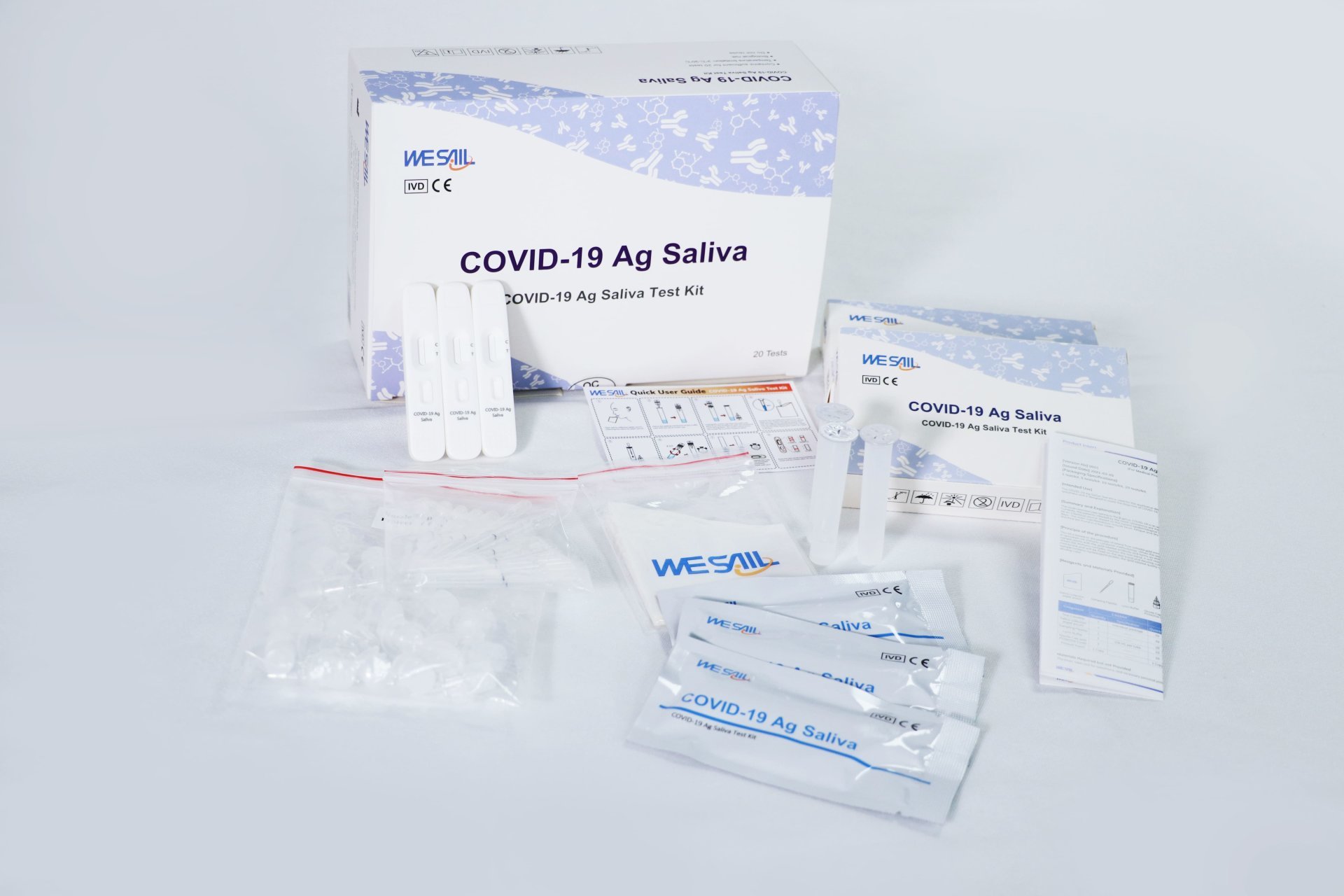
On April 10, 2021, the novel coronavirus (COVID-19) antigen saliva test kit (WESAIL COVID-19 antigen saliva test kit) developed and produced by Guangdong Weishi Biotechnology Co., Ltd. (WESAIL) has passed the expert review organized by the China Chamber of Commerce for Import and Export of Medicines and Health Products, and obtained permission from the Ministry of Commerce to enter the List of Medical Material Manufacturers Certified or Registered with Foreign Standards, which is the "White List" of the Ministry of Commerce for Export ", this means that the WESAIL New Crown Antigen Saliva Test Kit is officially eligible for export. Because the test kit has previously been CE certified by the European Union, after entering the "white list" of the Ministry of Commerce to comply with the procedures of the relevant importing countries can be exported to CE-recognized countries and regions, opened the pace of sales of WESAIL new crown antigen saliva test kit abroad.
WESAIL New Crown Antigen Saliva Test Kit can assist in the diagnosis and screening of infection during the incubation period, initial stage and infection period of virus infection. It is simple to operate, does not require equipment, is convenient to collect saliva samples, and can obtain diagnostic results within 15 minutes, eliminates sampling pain and fear, can better meet the needs of rapid detection on the scene of epidemic prevention and control in various countries, and will continue to assist in the prevention and control of novel coronavirus epidemic situation worldwide.
Recommended News



